How To Find Polarity Of A Molecule
How to find polarity of a molecule - The best way to explain this. The polarity of a molecule tells whether the electron cloud is equally distributed across the atoms within the molecule, or whether an electronegative atom is affecting the electron density.
Is SBr2 Polar or NonPolar (Sulfur Dibromide) in 2021
How do you determine the polarity of a molecule?
How to find polarity of a molecule - Decide whether the molecules represented by the following formulas are polar or nonpolar. Co 2 b.of 2 c. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; How to find polarity of a molecule
The distribution of the electrons will affect the behavior and reactivity of the molecule. Also, if the difference is amid 0.4 and 1.7 then the bond will appear polar. How to determine if a molecule is polar or not? How to find polarity of a molecule
Therefore, the shape of the molecule, and how electrons may be displaced across the molecule determine polarity. The terms “polar” and “nonpolar” usually refer to covalent bonds. Most cell components including proteins, polysaccharides and dna dissolve in water making it the basis of life. How to find polarity of a molecule
The steps on how to know if a molecule is polar or nonpolar may seem a bit. Molecular polarity the electron density of a polar bond accumulates towards one end of the bond, causing that end to carry a slight negative charge and the other end a slight positive charge. The polarity of bonds is caused due to the interaction of the bonds between molecules and atoms with different electronegativities. How to find polarity of a molecule
To determine the polarity of any molecule like h2s, it is equally important to find out its outside atoms, and shape. Determine which of the five categories of shapes your molecule falls. Start by drawing its lewis structure. How to find polarity of a molecule
Polarity is one of the properties of a compound related to other properties. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; Molecule polarity for molecules, polarity isn't quite so simple. How to find polarity of a molecule
Thus, to understand the properties of any molecule, one needs to explore its molecular structure which holds all the. Their bond polarity is determined according to the range it falls in: If the result is between 0.4 and 1.7, then, generally, the bond is polar covalent. How to find polarity of a molecule
If the result is between 0.4 and 1.7, then, generally, the bond is polar covalent. If the result is between 0.4 and 1.7, then, generally, the bond is polar covalent. The polarity makes water a universal solvent. How to find polarity of a molecule
(you may need to draw lewis structures and geometric sketches to do so.) a. Likewise molecules in which there is an accumulation of electron density at one end of the molecule, giving that end a partial negative charge and the other a partial positive charge, are. It can dissolve salts, acids, sugars, as well as alkalis and gases. How to find polarity of a molecule
The lewis structure will help you analyze the shape of the molecule given to you. In addition, for determining the polarity of a bond, you must find the difference of electronegativity of the atoms involved. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; How to find polarity of a molecule
For example, you can predict which solvents will be most effective with a given chemical if you know its. You'll need to take the difference between the electronegativity value of the two atom. But, if the difference is greater than this, then the bond will have an. How to find polarity of a molecule
This question can be easily answered by looking at the atoms and lone pair surrounding the central atom. Polarity refers to the physical properties of compounds such as boiling point, melting points and their solubilities. This rule applies to all molecules except hydrocarbons and molecules with two atoms of the same element. How to find polarity of a molecule
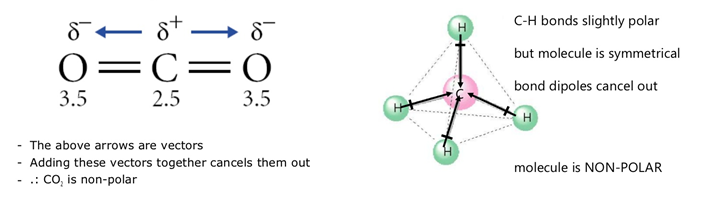
To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; This video looks at how to determine polarity in a molecule by understanding how the bond polarities, molecule shape, and outside atoms influence polarity us. We know that all bonds between dissimilar atoms are polar, but in a molecule, sometimes the dipole moments add up to form a net dipole moment of zero. How to find polarity of a molecule
How to determine if a molecule is polar or nonpolar. Consider an electromotive force (emf) or an electric potential, acting between two points. If the result is between 0.4 and 1.7, then, generally, the bond is polar covalent. How to find polarity of a molecule
How To Know If A Molecule Is Polar Or Nonpolar
3 Ways to Determine the Polarity of a Molecule wikiHow
How To Determine If A Molecule Is Polar Or Nonpolar
Unit 1 Elaboration Molecular Polarity
Examples on predicting molecular polarity YouTube
3 Ways to Determine the Polarity of a Molecule wikiHow
Ethanol Polar Or Nonpolar homework How do I figure out
Polar Molecules Tutorial How to determine polarity in a
Is Water Polar or Nonpolar Molecule? You Ask We Answer
Molecule Polarity Chemistry Tutorial YouTube
Stoichiometric Basics Chemistry for Kids! Graph for
Lesson Video Polar Bonding
Molecular Dipole The Overall Polarity of the Molecule
How to Determine if a Molecule is Polar or Not YouTube
Molecular Dipole The Overall Polarity of the Molecule
How To Find Net Dipole Moment
Ch4 Polar Or Nonpolar / Polar and NonPolar Molecules How
09 polarity 2016
Polar and Nonpolar Molecules YouTube